
May 4, 2021
Thermal properties and common applications of steel and other steel alloys
As currently the most sought-after metal globally, steel companies are continuously mass-producing this alloy to be used for a range of applications spanning multiple different industries. Its popularity can be attributed to its ability to form thousands of different compositions that display a unique combination of properties. These properties enable manufacturers to select a specific metal composition that is best suited to perform a specialized task.
On a basic level, steel can be described as an alloy of iron and carbon. An alloy is defined as a metal that is made by combining two or more metallic elements. In the case of steel, different alloys are formed to enhance the metal’s overall strength and increase its resistance to corrosion. Due to the extreme versatility of steel and its ability to be combined with a number of different elements, it has led to the creation of more than 3500 different grades of the metal that have been categorized based on their different properties. These grades are determined based on the amount of carbon present, how the metal has been processed, and other alloys that it may have been incorporated into the metal mixture.

Figure 1: Steel factory located in Europe. Photo from Tata Steel Europe.
Steel has one of the lowest thermal conductivity values of all metals, making it an ideal material to use in high-temperature environments such as vehicle or airplane engines. Thermal conductivity describes the rate at which thermal energy is transported through a material. This rate is measured in Watts per Meter per degrees Kelvin (W/(mK). A high thermal conductivity material can transport heat more quickly and efficiently than a material with low thermal conductivity.
Our experts are here to assist! Discover our services and advanced equipment, ensuring precise, ASTM and ISO-compliant measurements.
Poor thermal conductors, such as steel, carry heat very slowly and are ideal materials to use as insulators. Most metals display a high thermal conductivity and contain many fast-moving electrons, primarily responsible for conducting heat. The thermal conductivity of steel is measured at approximately 45 W/(mK), which is extremely low compared to copper and aluminum that exhibit a thermal conductivity value of 398 W/(mK) and 235 W/(mK) respectively.
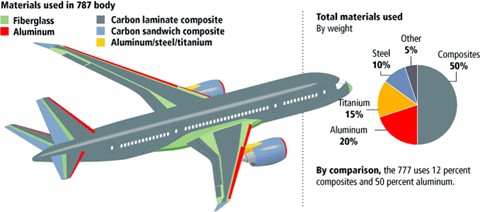
Figure 2: Diagram depicting the location of steel in an airplane. Ideal to use in areas with high exposer to heat such as in and around the engines. Photo from Aviation.Stack Exchange.
The versatility of steel and some of its key thermal properties have led to this metal dominating multiple industries. Steel is frequently used during the production of food processing equipment, medical devices, and kitchen appliances. Depending on the specific type of steel, its applications can become even more specific and specialized. Steel is often categorized into four groups, carbon steels, alloying steels, tool steels, and stainless steels.
Carbon steels contain only trace amounts of elements other than carbon and iron. This type of steel is the most commonly used, accounting for approximately 90% of steel production. Carbon steels have the highest thermal conductivity, averaging 45 Watts per Kelvin per meter. Heat treatment can harden steels containing over 0.1% carbon (depending on the thickness) to increase the metal’s strength. Manufacturers most frequently use annealing, hardening, and tempering as heat treatments.
We can classify carbon steel into three subgroups based on its carbon content. Low-carbon steels/mild steels contain up to 0.3% carbon and have low strength and high ductility, making carbon steel excellent for applications such as machine welding, structural shapes (l-beams, channels, and angle iron pipes), construction, bridge components, and food cans. Low-carbon steel is the most widely used subgroup and is a highly versatile and cost-effective option for production companies. Manufacturers often use medium carbon steel, which contains 0.3-0.6% carbon, for its superior strength and resistance to wear compared to low carbon steel. They commonly apply it to make machine parts, gears, railway tracks, and train wheels. High-carbon steel, containing more than 0.6% carbon, offers the highest wear resistance and strength. Manufacturers frequently use it to make cutting tools and springs.

Figure 3: Carbon (mild) steel rods. Photo: Jatinsanghvi/Commons.Wikimedia.org
Alloying steels incorporate additional elements, including nickel, copper, chromium and/or aluminum. The addition of these metals has a profound impact on the strength of the steel and other important properties such as ductility, corrosion resistance, and machinability. Tool steels are another main metal group that makes excellent cutting and drilling equipment as they contain tungsten, molybdenum, cobalt and vanadium. The addition of these elements can dramatically increase heat resistance and durability.
Stainless steel exhibits a low thermal conductivity of 15 W/(mK), which allows it to retain more energy that stabilizes the surrounding temperature better than other types of steel. Due to its ability to remain stable when exposed to higher temperatures, stainless steel is frequently used in applications such as food processing or ovens and conveyors that are frequently exposed to surges of high heat. The idea behind stainless steel was discovered in the early 19th century, but it took over 80 years to develop a reliable industrial method for producing the metal. There are currently over 150 grades of stainless steel; however, only 15 grades are frequently used.

Figure 4: Example of stainless-steel corroding. Photo: D3j4vu/Commons.Wikimedia.org
Stainless steel grades 304 and 316 are two of the most commonly manufactured types of the metal. Each type displays unique properties related to how they respond when exposed to different environments. Stainless steel 304 and 316 both contain a mixture of iron and chromium, but the exact ratio of the two metals is what produces a distinct difference between these two grades.
Stainless steel 304 is the most versatile and widely used grade of the metal and is ideal for applications that may be exposed to a higher temperature environment. This grade generally contains 18% chromium and 8% nickel. It is a key component in manufacturing sinks, saucepans, cutlery, tubing, brewery equipment, dairy, and food processing machines and pharmaceutical production equipment.
Stainless steel 316 contains less chromium (only 16%) but a greater amount of nickel and molybdenum. Although grade 316 comes in second in terms of quantity sold, it offers superior corrosion resistance to harmful chlorides and acids known to damage and stain steel. It is an extremely popular grade to use in medical equipment, implants, food service, processing and preparations, coastal environments, areas with high salt levels, and environments that experience increased exposure to alkalis and acids.
This increase in resistance to corrosion also makes cleaning easier as harmful chemicals are less likely to damage the steel’s appearance. If corrosion damage is not a major concern for construction companies, they will likely select grade 304 compared to 316 because it is a more cost-effective option.
Steel is a remarkably versatile metal that has a range of applications and uses spanning throughout multiple industries. Steel is an alloy that can be composed of various metals and elements that give it the ability to perform well under a variety of conditions when exposed to different environmental conditions.
All categories and grades of steel display an extremely low thermal conductivity, especially for metal. This gives steel the ability to maintain its structural integrity in environments that experience an increase in temperature and stress. These unique thermal properties and other key characteristics of steel are what makes and will continue to make this construction metal the most popular in the world.
Author: Kallista Wilson | Junior Technical Writer | Thermtest
304 vs 316 Stainless Steel: What You Need to Know. (2018, April 30). Retrieved August 26, 2020, from https://www.unifiedalloys.com/blog/304-316-stainless/
Carbon Steel: Properties, Production, Examples and Applications. (n.d.). Retrieved August 26, 2020, from https://matmatch.com/learn/material/carbon-steel
Comparing the Thermal Conductivity of Stainless Steel to other Metals. (2020, January 30). Retrieved August 26, 2020, from https://www.stainless-structurals.com/blog/comparing-the-thermal-conductivity-of-stainless-steel-to-other-metals/
Fussell, A. (2018, June 04). 10 Fun Facts About Stainless Steel. Retrieved August 26, 2020, from https://www.diversifiedmetals.com/10-fun-facts-stainless-steel/
Sponsored by Aalco – Ferrous and Non-Ferrous Metals StockistMay 18 2005. (2020, May 29). Stainless Steels – Stainless 304 Properties, Fabrication and Applications. Retrieved August 26, 2020, from https://www.azom.com/article.aspx?ArticleID=2867
STEELS. (n.d.). Retrieved August 26, 2020, from http://www.thermopedia.com/content/1152/
Thermal Properties of Carbon Steel – Melting Point … (n.d.). Retrieved August 26, 2020, from https://www.nuclear-power.net/nuclear-engineering/metals-what-are-metals/steels-properties-of-steels/carbon-steel-plain-carbon-steel/thermal-properties-of-carbon-steel-melting-point-thermal-conductivity/
Thermal Properties of Carbon Steel – Melting Point … (n.d.). Retrieved August 26, 2020, from https://www.nuclear-power.net/nuclear-engineering/metals-what-are-metals/steels-properties-of-steels/carbon-steel-plain-carbon-steel/thermal-properties-of-carbon-steel-melting-point-thermal-conductivity/